Health Canada has announced a nationwide recall of two lots of Seasonale birth control pills manufactured by Teva Canada Ltd. The recall was triggered by the discovery of packages containing an incorrect number of placebo pills, potentially reducing the medication’s effectiveness and increasing the risk of unintended pregnancy. This development highlights the critical importance of pharmaceutical quality control and consumer vigilance regarding prescription medications.
Contents
Key Takeaways:
- Teva Canada’s Seasonale birth control is being recalled.
- Affected packages contain extra placebo pills instead of active pills.
- This error can reduce the pill’s effectiveness, leading to unintended pregnancy.
- Two specific lots are impacted, identifiable by their numbers and expiry dates.
- Users should check their packages and consult a pharmacist or healthcare provider if affected.
What Happened: The Seasonale Recall Details
On June 4, Health Canada issued the recall for specific lots of Seasonale, a combination birth control pill containing levonorgestrel and ethinyl estradiol. The standard Seasonale package is designed with blister cards that include a specific sequence of active (pink) pills followed by a row of white placebo pills.
The recall was initiated after Teva Canada received a complaint that a package of Seasonale contained an extra row of white placebo pills in a section where only active pills should have been present. This manufacturing error means users might mistakenly take an inactive pill during a period when they should be receiving the hormonal medication required to prevent ovulation.
 Standard Seasonale birth control pill package showing active and placebo pills.
Standard Seasonale birth control pill package showing active and placebo pills.
Why This Matters: Implications for Effectiveness
The primary concern with taking extra placebo pills is the potential for reduced contraceptive efficacy. Birth control pills work by delivering precise doses of hormones at specific times in the menstrual cycle. Taking a placebo pill instead of an active hormonal pill disrupts this sequence, potentially allowing for ovulation to occur.
Health Canada explicitly stated in its advisory that “Taking placebo pills instead of active (pink) pills may reduce the effectiveness of the product and could lead to an unintended pregnancy.” This underscores the serious health implication of the packaging error for individuals relying on Seasonale for contraception. Other side effects, such as spotting and irregular bleeding, can also occur when pills are taken out of their intended order.
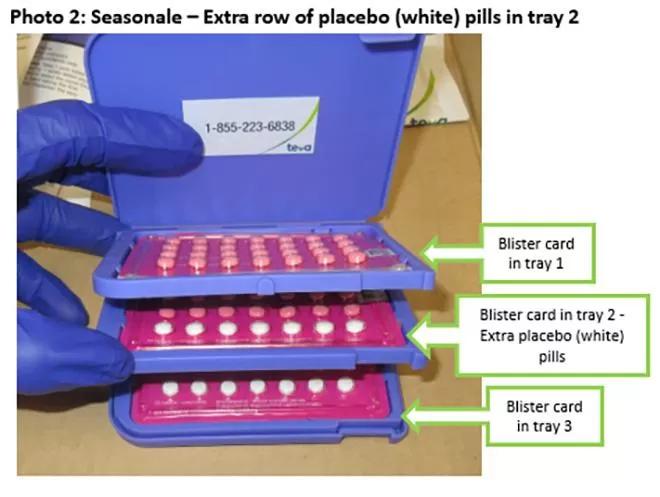 Recalled Seasonale birth control package displaying an extra row of white placebo pills.
Recalled Seasonale birth control package displaying an extra row of white placebo pills.
Identifying Affected Lots and Required Action
The recall specifically affects two lots of Seasonale birth control pills:
- Lot Number: 100059661
- Expiration Date: January 31, 2026
- Lot Number: 100069150
- Expiration Date: September 30, 2026
Canadians using Seasonale are urged to immediately check their package and blister cards to confirm if they have pills from these specific lots.
If your package is part of the affected lots:
- Do not take any more pills from that package.
- Return the product to your pharmacy for a replacement or an alternative birth control product.
- If you cannot get to a pharmacy immediately, identify the next active (pink) pill in the proper sequence according to the package instructions and take it. Contact your pharmacist as soon as possible.
- If you have already taken a placebo pill incorrectly or are unsure, use non-hormonal backup contraception (like condoms).
- Consult with a healthcare professional (like a doctor or pharmacist) to discuss your situation, review your birth control options, and address any concerns about potential pregnancy or side effects.
Regulatory Oversight and Company Response
Health Canada is actively monitoring the recall process and Teva Canada’s investigation into the root cause of the packaging error. The regulatory body is also overseeing the steps the company is implementing to prevent similar incidents from happening in the future. Pharmaceutical companies are required to adhere to strict quality control standards, and errors leading to product recalls trigger scrutiny from health authorities.
 Symbolic image representing a product recall in Canada.
Symbolic image representing a product recall in Canada.
Context: Recent Quality Control Issues
This is not the first time Teva Canada has faced a recall for its birth control products this year. In May, Health Canada issued a recall for Seasonique, another birth control pill by Teva Canada, after a complaint that a package was missing two pills. While the specific errors differ (extra placebos versus missing active pills), these instances collectively raise questions about the company’s quality control processes for its contraceptive products in recent months.
What’s Next: Ensuring Safety and Effectiveness
For consumers, the immediate next step is to verify their specific product lot and take appropriate action based on Health Canada’s guidance. For Teva Canada, this recall necessitates a thorough review and potential overhaul of their packaging and quality assurance procedures to restore confidence in their products. Health Canada’s ongoing monitoring will ensure compliance and help prevent future errors that could compromise patient safety and the effectiveness of essential medications. Users should remain vigilant and consult healthcare providers with any concerns about their medication.







































