Weight loss medications like Ozempic and Wegovy have been a game-changer for many, helping with weight management and improving health markers by mimicking natural appetite-regulating hormones. While incredibly effective, they often come with uncomfortable side effects, most notably nausea and vomiting. But what if you could get the benefits without the sickness?
Contents
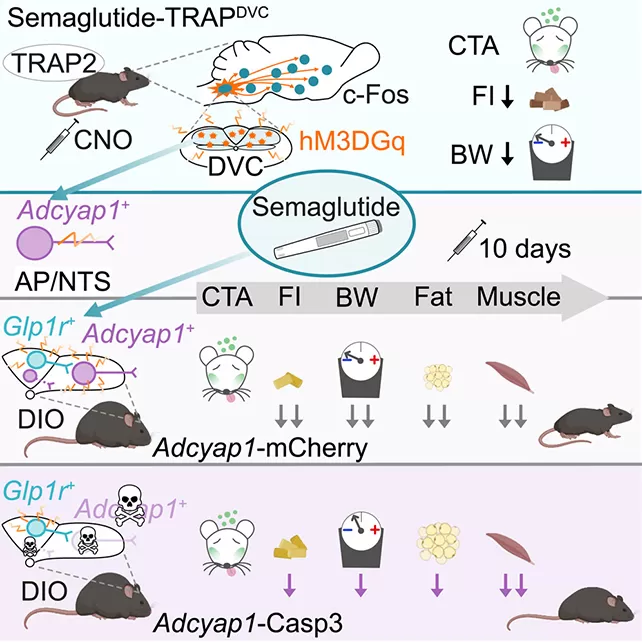
New research from the University of Gothenburg in Sweden has pinpointed a specific group of nerve cells in the brains of mice that appear linked only to the positive effects of semaglutide (the active ingredient in these drugs) – the appetite suppression and weight loss – while seeming to avoid the unpleasant side effects. This breakthrough could pave the way for future weight loss treatments that are much easier to tolerate.
How Semaglutide Works (and the Side Effect Puzzle)
Medications like Ozempic and Wegovy belong to a class called GLP-1 receptor agonists. Think of GLP-1 as a natural hormone your body produces after you eat; it signals your brain that you’re full and helps manage blood sugar. Semaglutide activates the same receptors, essentially boosting this “I’m full” signal. This is why the drugs are so effective at reducing appetite and promoting weight loss.
However, these GLP-1 receptors are found in many places throughout the body and brain. Activating them broadly can lead to various effects, including those beneficial ones and the unwelcome side effects like nausea, which is often linked to GLP-1 activity in areas of the brain that control vomiting, and potential loss of muscle mass.
Discovering the Key Neurons in the Brain
Researchers wanted to understand if specific brain cells were responsible for the weight loss effects separately from the side effects. They administered semaglutide to mice and carefully observed which neurons in the brain became active.
Their search led them to a particular set of neurons, technically called Adcyap1+ neurons, located in a part of the brainstem known as the dorsal vagal complex. This area is crucial for processing signals from the body’s organs, including the gut.
Pinpointing the “Good” Effects
To test the role of these specific Adcyap1+ neurons, the scientists conducted further experiments:
- They artificially stimulated these particular neurons in mice. The result? The mice ate less and lost weight, similar to the effects of semaglutide, but without showing signs of nausea.
- They genetically disabled or destroyed these specific neurons in another group of mice. When these mice were then given semaglutide, the drug’s ability to suppress appetite and cause weight loss was significantly reduced. Crucially, the side effects like nausea still seemed to occur.
This strongly suggests that these Adcyap1+ neurons are primarily responsible for the positive effects of semaglutide on appetite and weight loss, while other neural pathways or parts of the body might be driving the side effects.
What This Means for Future Treatments
Identifying these specific neurons is a huge step forward. It opens the door to the possibility of developing new therapies that could specifically target only these beneficial neurons.
“If we can target the treatment there,” says neuroscientist Júlia Teixidor-Deulofeu from the University of Gothenburg, “we may be able to maintain the positive effects while reducing side effects.”
Imagine a future weight loss treatment that offers the effectiveness of today’s leading drugs but is much more comfortable to take, free from the debilitating nausea that many users experience.
A Step Towards Smarter Medicine
It’s important to remember that this research was conducted in mice, and potential treatments for humans are still a long way off. However, this study provides invaluable insight into exactly how semaglutide works in the brain to achieve its weight loss effects.
Currently, semaglutide is being explored for treating various conditions beyond weight loss and diabetes, including substance use disorders and neurodegenerative diseases. A deeper understanding of its mechanisms in the brain, like the role of these specific neurons, allows researchers to refine its use and potentially develop better versions of the drug or entirely new therapies.
“It is important to understand how these drugs actually work,” adds neuroscientist Linda Engström Ruud. “The better we understand this, the greater the opportunity we have to improve them.”
This research brings us closer to understanding the complex interplay between the gut, brain, and our metabolism, potentially leading to more effective and tolerable weight loss solutions in the future.
The findings were published in the journal Cell Metabolism.