Cancer is a complex disease, often driven by tiny changes in our cells’ machinery. One key player frequently involved is a protein called BRAF. Now, exciting new research sheds light on how mutant forms of BRAF cause cancer and, crucially, reveals a groundbreaking way to potentially stop them. This study, led by a team at Université de Montréal’s IRIC, published in Science, offers fresh hope for developing more effective cancer treatments.
Contents
Here’s the big picture:
- About half of all cancers involve problems with a cellular communication system called the MAPK pathway.
- BRAF acts like a central ‘on/off’ switch in this system, normally kept tightly controlled.
- Mutations in BRAF can make it stuck in the ‘on’ position, leading to uncontrolled cell growth.
- Researchers discovered that mutant BRAF achieves this by changing its shape to mimic its active form, essentially tricking the cell.
- Even better: They found small molecules that can force this rogue, mutated BRAF back into its correct, ‘off’ shape.
The BRAF Problem: A Molecular Switch Gone Rogue
Imagine your cells have a sophisticated internal phone system (the MAPK pathway) that helps them respond to signals from the outside world. BRAF is a critical operator in this system, acting like a switch that needs to be flipped “on” only when necessary. Normally, BRAF has built-in controls that keep it in an “off” state – think of it as a safety lock called “auto-inhibition.”
However, certain genetic mistakes, known as oncogenic mutations, can affect the BRAF protein. These mutations are commonly found in various cancers, including melanoma (skin cancer), thyroid, colon, and lung cancers. When BRAF is mutated, it can bypass its normal safety lock and become constantly “on,” leading to the cell receiving non-stop growth signals. This uncontrolled growth is a hallmark of cancer.
For a long time, scientists knew these mutations were causing the problem, but precisely how the altered BRAF protein managed to escape control wasn’t fully understood.
Unlocking the Secret: A Deceptive New Shape
Using advanced imaging techniques, specifically cryo-electron microscopy (which lets scientists see the detailed 3D structure of tiny proteins), the Université de Montréal team, led by Professor Marc Therrien, peered into the world of mutated BRAF.
What they discovered was fascinating: the mutated forms of BRAF don’t just break the safety lock; they adopt a structural shape that looks almost identical to BRAF’s naturally active state. It’s like the mutated protein puts on a perfect disguise, fooling the cell’s internal checkpoints into thinking it’s supposed to be active. This allows the rogue BRAF to trigger signaling pathways without restriction, driving unlimited cell proliferation.
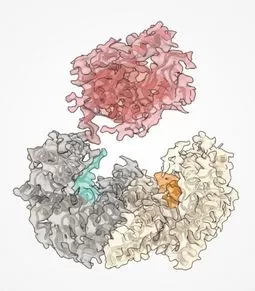 Representation of the BRAF protein structure in salmon color. Other proteins (chaperones) are shown helping it fold.
Representation of the BRAF protein structure in salmon color. Other proteins (chaperones) are shown helping it fold.
They pinpointed a specific segment of the protein called the alpha-C helix. In healthy BRAF, this helix is positioned in a way that helps keep the protein inactive. But in the mutated, cancer-causing forms, this alpha-C helix shifts its position to match where it would be in a normally active protein. This subtle structural change is key to its deceptive behavior.
A Potential Solution: Forcing the Switch Back ‘Off’
Understanding how mutant BRAF evades control is a major step, but the research didn’t stop there. The team then explored whether they could use small drug-like molecules to target this crucial alpha-C helix and influence its position.
In a significant breakthrough, some of the molecules they tested were able to interact with the mutated BRAF protein in a way that forced the alpha-C helix back into its ‘inactive’ position. This effectively reverted the hyperactive, cancer-driving form of BRAF back to its normal, inactive, auto-inhibited state.
This is a powerful finding. Most BRAF-targeting drugs currently work by blocking the downstream signal after BRAF is already active. This new approach suggests it might be possible to tackle the problem earlier by forcing the mutant protein itself back into a harmless shape before it can even start signaling.
The ability to completely change the shape of a mutant protein using a therapeutic molecule is a first in this specific field of research and opens up exciting possibilities.
What This Means for Future Cancer Treatments
This study provides crucial insights into the fundamental mechanics of how BRAF mutations contribute to cancer. By understanding the specific structural changes and the role of the alpha-C helix, scientists can now work on designing and optimizing entirely new classes of drugs.
Instead of just blocking the effects of rogue BRAF, future therapies could potentially be developed to specifically target and “reconvert” these mutated proteins, forcing them back into an inactive state. This could be a more precise and effective way to neutralize BRAF-driven cancers. While this research is a foundational step and more work is needed, it paves a promising new path for drug discovery and offers hope for better treatments for patients affected by BRAF-mutant cancers.
For more on cancer research and related topics, explore these resources: [Link to relevant research section 1], [Link to relevant research section 2].